|
Foodlaw-Reading
Dr David Jukes, The University of
Reading, UK
Providng access to food law since May 1996 |

|
.....  ..... ..... ..... .....  ..... .....  ..... .....  ..... ..... 
|
Last updated:
1 January, 2021
Novel Foods - Archive Page
Providing access to former EU Legislation and the process of adopting the current controls. For the current controls, see: Novel Foods.
Former EU Law
The original regulation was:
- Regulation (EC) No 258/97 of the European Parliament and of the Council of 27 January 1997 concerning novel foods and novel food ingredients
Amendments:
- Regulation (EC) No 1829/2003 of the European Parliament and of the Council of 22 September 2003 on genetically modified food and feed (OJ L268, 18.10.2003, page 1) [Removal of GM food from Regulation]
- Regulation (EC) No 1882/2003 of the European Parliament and of the Council of 29 September 2003 adapting to Council Decision 1999/468/EC the provisions relating to committees which assist the Commission in the exercise of its implementing powers laid down in instruments subject to the procedure referred to in Article 251 of the EC Treaty (OJ L284, 31.10.2003, page 1) [Amendments to Committee procedures]
- Regulation (EC) No 1332/2008 of the European Parliament and of the Council of 16 December 2008 on food enzymes and amending Council Directive 83/417/EEC, Council Regulation (EC) No 1493/1999, Directive 2000/13/EC, Council Directive 2001/112/EC and Regulation (EC) No 258/97 (OJ L354, 31.12.2008, page 7) [Excluded enzymes from the scope of the Regulation
Consolidated Text: A consolidated version of the final text of the Regulation incorporating these amendments is available. See: Regulation 258/1997 - Consolidated text (January 2009)
For more details about the legislation, supporting documents, approvals and procedures see:
For a register of novel foods approved under this former Regulation, see the Commission's page at: https://ec.europa.eu/food/safety/novel_food/authorisations/list_authorisations_en
Development of the new EU Controls
In December 2007 the Commission adopted a proposal for updating the novel food legislation. This was published in January 2008. However there was no agreement between the Institutions - particularly the Council and the European Parliament. Ultimately, despite conciliation, the proposal was not adopted and lapsed in March 2011. The main areas of disagreement related to nanotechnology and animal cloning. The Commission published revised proposals in December 2013. This included one which would replace the original novel food regulation and two others specifically on animal cloning. Although there has been little progress on the issue of animal cloning, the revised regulation on novel foods was adopted in November 2015.
First Proposal (2007-2011)
- COM (2007) 0872- Proposal for a Regulation of the European Parliament and of the Council on novel foods and amending Regulation (EC) No XXX/XXXX (common procedure)
This figure shows the EU procedure for adopting legislation. The coloured items refer to stages which were followed. It can be seen that in March 2011, the discussions concluded without the adoption of new controls. The work ceased and the Commission had to bring forward new proposals to start the whole procedure again.
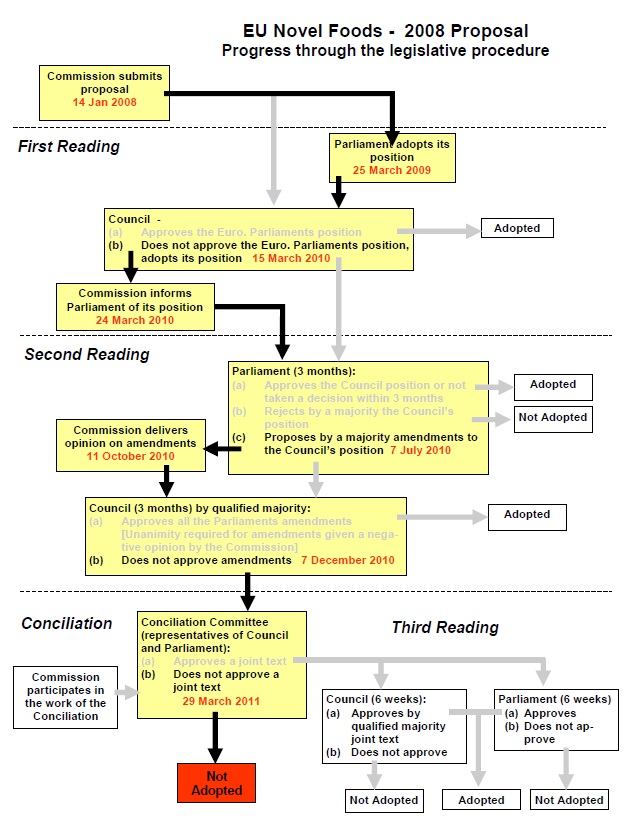
Click here for full size version (pdf file)
Key documents:
- European Parliament legislative resolution of 25 March 2009 on the proposal for a regulation of the European Parliament and of the Council on novel foods and amending Regulation (EC) No XXX/XXXX
- Position (EU) No 6/2010 of the Council at first reading with a view to the adoption of a Regulation of the European Parliament and of the Council on novel foods, amending Regulation (EC) No 1331/2008 and repealing Regulation (EC) No 258/97 and Commission Regulation (EC) No 1852/2001 Adopted by the Council on 15 March 2010 (OJ C122E, 11.5.2010, page 38)
- Communication from the Commission to the European Parliament pursuant to Article 294(6) of the Treaty on the Functioning of the European Union concerning the position of the Council on the adoption of a Regulation of the European Parliament and of the Council on novels foods, amending Commission Regulation (EC) No 1331/2008 and repealing Regulation (EC) No 258/97 and Commission Regulation (EC) No 1852/2001 (COMDOC(2010)0124)
- Opinion of the Commission pursuant to Article 294, paragraph 7, point (c) of the Treaty on the Functioning of the European Union, on the European Parliament's amendments to the Council's position regarding the proposal for a REGULATION OF THE EUROPEAN PARLIAMENT AND OF THE COUNCIL on novel foods, amending Regulation (EC) N° 1331/2008 and repealing Regulation (EC) N° 258/97 and Commission Regulation (EC) N° 1852/2001 (COM (2010) 0570)
News items:
Key links to relevant news items are given here:
- 29 March 2011 Statement by Commissioner Dalli on the lack of agreement in the conciliation procedure on the Novel Food Regulation
- 29 March 2011 Conciliation on novel foods failed
- 7 December 2010 Council rejects the Parliament’s Proposed Amendments to the Draft Regulation
- 11 October 2010 Commission issues its opinion on Parliament's proposed amendments (see COM (2010) 0570)
- 7 July 2010 MEPs call for ban on food from cloned animal
- 4 May 2010 MEPs vote to exclude food from cloned animals
- 10 May 2010 Article from the European Parliament: risk assessment for nano-foods
- 15 March 2010 Council adopts its position on novel food
- 22 June 2009 Council agrees on new rules for novel foods
- 25 March 2009 MEPs adopt report on proposed new rules
- 28 April 2008 FSA consultation on proposal for a European regulation on novel foods
- 14 January 2008 Commission proposes measures to encourage safe novel foods in the EU
- 14 January 2008 Questions and Answers on Novel Foods
Second Proposal (2013-2015)
At this second attempt, there was a much greater willingness to reach compromise. Detailed discussions were held between the European Parliament and the Council to achieve an agreed document. As shown in this diagram, the discussions led to an agreed text such that the amendments adopted by the European Parliament at First Reading were all accepted by the Council:
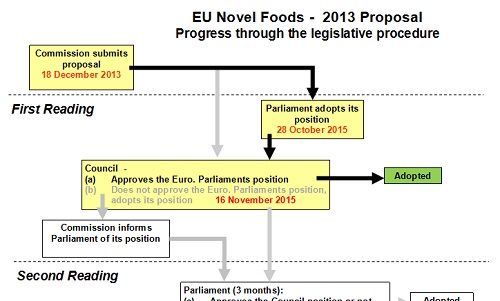
Click here for full size version (pdf file)
News Items:
Key links to relevant news items are given here:
- 3 January 2018 New regulation adding to the food variety present on the EU market enters into force
- 5 October 2017 Commission consultation: Draft Regulation establishing the Union list of novel foods in accordance with Regulation (EU) 2015/2283 of the European Parliament and of the Council on novel foods
- 19 July 2017 Commission Implementing Regulation laying down administrative and scientific requirements for novel food applications
- 19 July 2017 Draft implementing regulation: Commission Impl. Reg. laying down administrative & scientific requirements for traditional foods from a Third Country
- 19 July 2017 Draft implementing regulation: Implementing act on procedures for novel food status
- 10 November 2016 Novel and traditional food: guidance finalised by EFSA
- 16 November 2015 New novel food Regulation has been agreed today: Questions and Answers: New Regulation on Novel Food
- 16 November 2015 Commissioner for Health and Food Safety Vytenis Andriukaitis welcomes the political agreement on novel food
- 11 November 2015 New rules on novel foods get Council's approval
- 28 October 2015 Novel foods: simpler approval rules to boost food innovation
- 22 October 2015 Speeding up authorisation of novel foods: European Parliament provide background information
- 8 September 2015 EP wants animal cloning ban extended to offspring and imports
- 16 July 2015 Nanomaterial definition: final European Commission report
- 25 June 2015 Food safety MEPs back deal with Council
- 10 June 2015 Council presents final compromise text
- 12 May 2015 EU talks go on: Council and Parliament discuss proposed legislation
- 25 November 2014 MEPs call for moratorium on nano-foods and labelling of cloned meat
- 13 March 2014 Parliament Discussions: Defining nano-food: a big problem at a very small scale
- 12 March 2014 Parliament Discussions: Nanofoods: MEPs object to new labelling rules
- 18 December 2013 Food: Commission tables proposals on animal cloning and novel food
This page was first provided on 1 January 2021 by transferring material from the main Novel Foods page.
To go to main Foodlaw-Reading
Index page, click here.
![]()
