|
Click here for a general introduction to our work. Suitable for the general public. Click here for more detailed information on our research interests. Suitable for scientists, clinicians and students. Funded by: |
Apoptosis and nitric oxide
Apoptosis, or programmed cell death, is a normal component of the development and health of multicellular organisms. Cells die in response to a variety of stimuli and during apoptosis they do so in a controlled, regulated fashion. This makes apoptosis distinct from another form of cell death called necrosis in which uncontrolled cell death leads to lysis of cells, inflammatory responses and, potentially, to serious health problems. Apoptosis, by contrast, is a process in which cells play an active role in their own death (which is why apoptosis is often referred to as cell suicide).
Upon receiving specific signals instructing the cells to undergo apoptosis a number of distinctive changes occur in the cell. A family of proteins known as caspases are typically activated in the early stages of apoptosis. These proteins breakdown or cleave key cellular components that are required for normal cellular function including structural proteins in the cytoskeleton and nuclear proteins such as DNA repair enzymes. The caspases can also activate other degradative enzymes such as DNases, which begin to cleave the DNA in the nucleus.
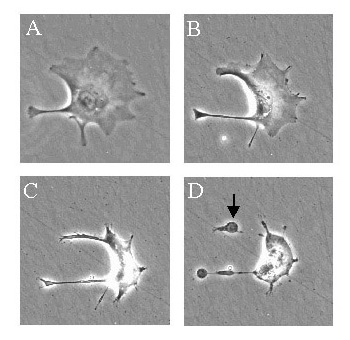 Apoptotic cells display distinctive morphology during the apoptotic process. This can be seen in the image above, taken using a scanning electron microscope, and also in the image on the right and the movie below which shows a trophoblast cell undergoing apoptosis.
Apoptotic cells display distinctive morphology during the apoptotic process. This can be seen in the image above, taken using a scanning electron microscope, and also in the image on the right and the movie below which shows a trophoblast cell undergoing apoptosis.
Typically, the cell begins to shrink following the cleavage of lamins and actin filaments in the cytoskeleton (A). The breakdown of chromatin in the nucleus often leads to nuclear condensation and in many cases the nuclei of apoptotic cells take on a "horse-shoe" like appearance (B). Cells continue to shrink (C), packaging themselves into a form that allows for their removal by macrophages. These phagocytic cells are responsible for clearing the apoptotic cells from tissues in a clean and tidy fashion that avoids many of the problems associated with necrotic cell death. In order to promote their phagocytosis by macrophages, apoptotic cells often ungergo plasma membrane changes that trigger the macrophage response. One such change is the translocation of phosphatidylserine from the inside of the cell to the outer surface. The end stages of apoptosis are often characterised by the appearance of membrane blebs (D) or blisters process. Small vesicles called apoptotic bodies are also sometimes observed (D, arrow).
There are a number of mechanisms through which apoptosis can be induced in cells. The sensitivity of cells to any of these stimuli can vary depending on a number of factors such as the expression of pro- and anti-apoptotic proteins (eg. the Bcl-2 proteins or the Inhibitor of Apoptosis Proteins), the severity of the stimulus and the stage of the cell cycle. Some of the major stimuli that can induce apoptosis are outlined in the illustration below.
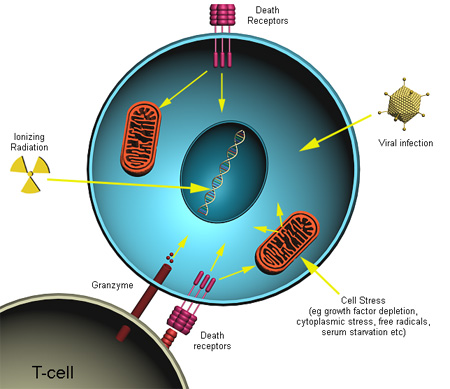
In some cases the apoptotic stimuli comprise extrinsic signals such as the binding of death inducing ligands to cell surface receptors called death receptors. These ligands can either be soluble factors or can be expressed on the surface of cells such as cytotoxic T lymphocytes. The latter occurs when T-cells recognise damaged or virus infected cells and initiate apoptosis in order to prevent damaged cells from becoming neoplastic (cancerous) or virus-infected cells from spreading the infection. Apoptosis can also be induced by cytotoxic T-lymphocytes using the enzyme granzyme.
In other cases apoptosis can be initiated following intrinsic signals that are produced following cellular stress. Cellular stress may occur from exposure to radiation or chemicals or to viral infection. It might also be a consequence of growth factor deprivation or oxidative stress caused by free radicals. In general intrinsic signals initiate apoptosis via the involvement of the mitochondria. The relative ratios of the various bcl-2 proteins can often determine how much cellular stress is necessary to induce apoptosis.
The caspases are a family of proteins that are one of the main executors of the apoptotic process. They belong to a group of enzymes known as cysteine proteases and exist within the cell as inactive pro-forms or zymogens. These zymogens can be cleaved to form active enzymes following the induction of apoptosis.
Induction of apoptosis via death receptors typically results in the activation of an initiator caspase such as caspase 8 or caspase 10. These caspases can then activate other caspases in a cascade. This cascade eventually leads to the activation of the effector caspases, such as caspase 3 and caspase 6. These caspases are responsible for the cleavage of the key cellular proteins, such as cytoskeletal proteins, that leads to the typical morphological changes observed in cells undergoing apoptosis.
The apoptosome
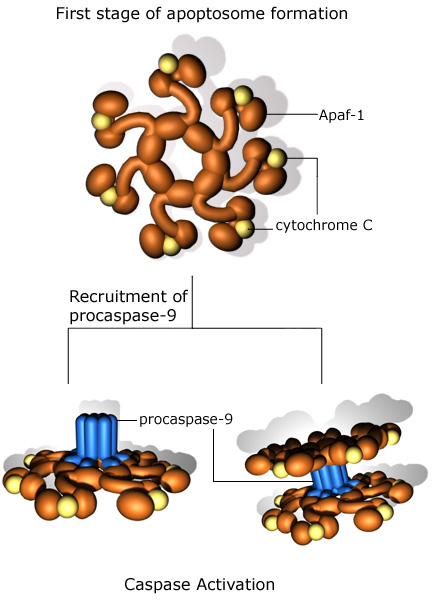
There are a number of other mechanisms, aside from activation of the death receptors, through which the caspase cascade can be activated. Granzyme B can be delivered into cells by cytotoxic T lymphocytes and is able to directly activate caspases 3, 7, 8 and 10. The mitochondria are also key regulators of the caspase cascade and apoptosis. Release of cytochrome C from mitochondria can lead to the activation of caspase 9, and then of caspase 3. This effect is mediated through the formation of an apoptosome, a multi-protein complex consisting of cytochrome C, Apaf-1, pro-caspase 9 and ATP. The formation of the apoptosome is illustrated below.
Caspases and chromatin breakdown
One of the hallmarks of apoptosis is the cleavage of chromosomal DNA into nucleosomal units. The caspases play an important role in this process by activating DNases, inhibiting DNA repair enzymes and breaking down structural proteins in the nucleus. This processes is illustrated below.
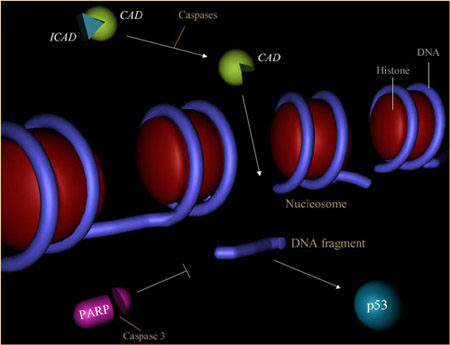
1) Inactivation of enzymes involved in DNA repair.
The enzyme poly (ADP-ribose) polymerase, or PARP, was one of the first proteins identified as a substrate for caspases. PARP is involved in repair of DNA damage and functions by catalyzing the synthesis of poly (ADP-ribose) and by binding to DNA strand breaks and modifying nuclear proteins. The ability of PARP to repair DNA damage is prevented following cleavage of PARP by caspase-3.
2) Breakdown of structural nuclear proteins.
Lamins are intra-nuclear proteins that maintain the shape of the nucleus and mediate interactions between chromatin and the nuclear membrane. Degradation of lamins by caspase 6 results in the chromatin condensation and nuclear fragmentation commonly observed in apoptotic cells.
3) Fragmentation of DNA.
The fragmentation of DNA into nucleosomal units - as seen in DNA laddering assays - is caused by an enzyme known as CAD, or caspase activated DNase. Normally CAD exists as an inactive complex with ICAD (inhibitor of CAD). During apoptosis, ICAD is cleaved by caspases, such as caspase 3, to release CAD. Rapid fragmentation of the nuclear DNA follows.
Death Receptor Signalling
Death receptors are cell surface receptors that transmit apoptotic signals initiated by specific ligands such as Fas ligand, TNF alpha and TRAIL. They play an important role in apoptosis and can activate a caspase cascade within seconds of ligand binding. Induction of apoptosis via this mechanism is therefore very rapid.
Apoptotic signalling from the death receptors
Although there are differences in the signalling pathways activated by the different death receptors it is possible to outline a general apoptotic signalling pathway, as seen in the animation below.
Binding of the death inducing ligand to its receptor can lead to a the generation of ceramide, typically produced by acid sphingomyelinase. This ceramide release is thought to promote lipid raft fusion which results in a large scale clustering of the death receptors. The large scale receptor clustering is important because it helps amplify the apoptotic signalling. In the absence of receptor clustering some cells, such as lymphocytes, are still able to trigger apoptosis but in most cases amplification of the signalling pathway is needed to activate the full apoptotic response.
Following ligand binding a conformational change in the intracellular domains of the receptors reveals the presence of a "death domain" which allows the recruitment of various apoptotic proteins to the receptor. This protein complex is often called the DISC, or Death Inducing Signalling Complex. The final step in this process is the recruitment of one of the caspases, typically caspase 8, to the DISC. This results in activation of caspase 8 and the inititation of apoptosis.
TNF receptor signalling
TNF is produced by T-cells and activated macrophages in response to infection. By activating its receptor, TNFR1, TNF can have several effects (see illustration below).
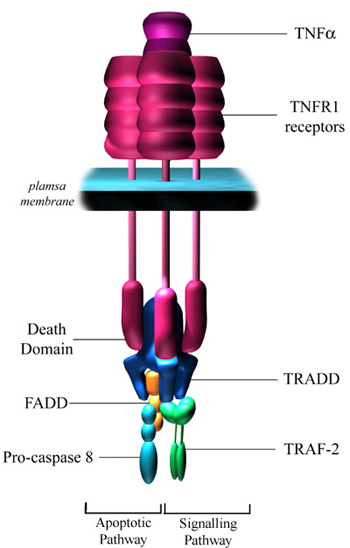 In some cells it leads to activation of NF-kB and AP-1 which leads to the induction of a wide range of genes. In some cells, however, TNF can also induce apoptosis, although receptor ligation is rarely enough on its own to initiate apoptosis as is the case with Fas ligand binding.
In some cells it leads to activation of NF-kB and AP-1 which leads to the induction of a wide range of genes. In some cells, however, TNF can also induce apoptosis, although receptor ligation is rarely enough on its own to initiate apoptosis as is the case with Fas ligand binding.
Binding of TNF alpha to TNFR1 results in receptor trimerisation and clustering of intracellular death domains. This allows binding of an intracellular adapter molecule called TRADD (TNFR-associated death domain) via interactions between death domains. TRADD has the ability to recruit a number of different proteins to the activated receptor. Recruitment of TRAF2 (TNF-associated factor 2) can lead to activation of NF-kB and the JNK pathway.
TRADD can also associate with FADD, which leads to the induction of apoptosis via the recruitment and cleavage of pro-caspase 8.
Signaling by Fas (CD95)
The ligand for Fas (FasL or CD95L) activated apoptosis in a similar way to the TNF receptor. Binding of the ligand promotes receptor clustering, DISC formation and the activation of the caspase cascade. However, signalling through the Fas receptor is slightly simpler than through the TNF receptor. The adapter protein FADD can be recruited directly to the death domain on the Fas receptor, without requiring the prior recruitment of TRADD. In addition the Fas receptor is generally though to only activate apoptosis and does not play an important role in other aspects of cell signalling like the TNF receptor.
Induction of apoptosis by TRAIL
In a number of ways TRAIL (TNF-related apoptosis inducing ligand) is similar in action to FasL. Binding of TRAIL to its receptors DR4 or DR5 triggers rapid apoptosis in many cells. Interestingly there are also decoy receptors that compete for binding of TRAIL with the DR4 and DR5 receptors. The decoy receptors are called DcR1 and DcR2. Both of these receptors are capable of competing with DR4 or DR5 receptors for binding to the ligand (TRAIL), however ligation of these receptors does not initiate apoptosis since DcR1 does not possess a cytoplasmic domain, while DcR2 has a truncated death domain lacking 4 out of 6 amino acids essential for recruiting adapter proteins.
![]()
![]()




